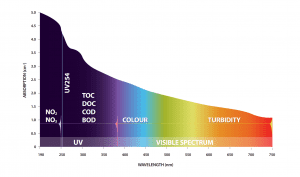
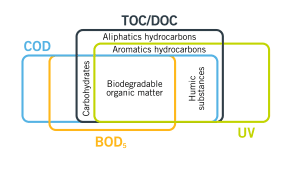
UV254, also known as the Spectral Absorption Coefficient (SAC), is a water quality test parameter which utilizes light at the UV 254nm wavelength to be able to detect organic matter in water and wastewater. This is due to the fact that most organic compounds absorb light at the UV 254 nm wavelength.
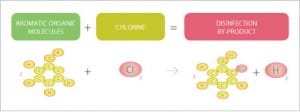
Unlike other organic test parameters, UV254 has a bias towards reactive or aromatic organic matter which has double bonded ring structures and is typically the most problematic form of organics in water.
UV254 is typically represented as a calculation of UV absorbance (UVA) or UV transmittance (UVT).